BasicBites®–
A Revolutionary Oral Microbiome Approach to Boost Enamel Health
Available as a downloadable PDF.
Tooth decay remains the most common prevalent disease in children and adults. The most critical contributing factor in the development of caries is a low pH which indicates the oral environment is acidic. Most conventional oral care technologies (e.g., fluoride), however, do not address this primary source of caries. They are mainly defensive measures designed to reduce demineralization and support remineralization which helps teeth fight off the effects of harmful, acid-producing bacteria. Although antimicrobial products such as certain mouthwashes claim to eradicate “bad” bacteria, they also may destroy virtually all the “good” bacteria necessary to maintain a balanced pH — the key to a healthy oral microbiome and essential for enamel health.
The breakthrough arginine bicarbonate, calcium carbonate microbiome technology of BasicBites® takes a different approach to supporting enamel health. By nurturing and activating the beneficial alkali generating bacteria discovered in biofilms, BasicBites creates an optimal oral environment that supports not only a pH-balanced microbial ecosystem, but also the protection and remineralization of tooth enamel. Unlike other oral care products, BasicBites plays both offense and defense in a prebiotic/postbiotic process that delivers immediate and sustained neutralization of plaque acids. This is accomplished without killing bacteria or adding new living microbes which may never adapt to the oral microbial ecosystem. Its unique technology also has been clinically shown to be effective in plugging and sealing open dentinal tubules to restrict movement of inter-tubular fluid that can adversely impact dentinal hypersensitivity.
The breakthrough arginine bicarbonate and calcium carbonate oral microbiome technology of BasisBites offers all-in-one enamel protection in a tasty, 15-calorie, sugar-free soft chew. Numerous clinical and research studies have validated the beneficial effects of this technology, including:
• A one-year, double-blind clinical investigation found that BasicBites reduced the progression of non-cavitated occlusal caries lesions in the first permanent molars of children by 42%.(1)
• A study of the effects of a fluoride-free arginine bicarbonate/calcium carbonate in toothpaste used by 11 to 12-year-old children for two years found that it was significantly more effective in inhibiting caries initiation and progression than the fluoride toothpaste used by the control group.(2)
• The authors of one study noted that “breakthrough technology based upon arginine and calcium carbonate provides clinically proven benefits with respect to rapid and lasting relief of dentin hypersensitivity.”(3)
• In another clinical evaluation of an arginine bicarbonate/calcium carbonate complex in a sugar-free mint used by 200 10 to 11-year-old children, its authors concluded that this technology offers, “a simple and economical means for reducing substantially one of the most prevalent diseases in these children.”(4)
Our Origin Story
The revolutionary enamel-protecting microbiome technology in BasicBites was pioneered by Israel Kleinberg DDS, PhD, DSc, who is widely considered to be the “Grandfather” of modern oral biology. The oral microbiome harbors billions of microorganisms including hundreds of species of bacteria. For decades, scientists have known that the main source of tooth decay is cariogenic oral bacteria which metabolize simple and complex sugars to produce acids that cause the dissolution of tooth enamel.
Kleinberg’s early research focusing on a mixed-bacterial/ecological approach to understand the role of bacteria in dental caries causation led to saliva fractionation studies that identified arginine, an amino acid, as a key component responsible for the pH-raising effect of saliva.(5) Continued studies in his lab led to the discovery of numerous commensal oral bacteria that could metabolize arginine and generate alkali as a byproduct.(6)
By combining arginine with calcium and carbonate/ bicarbonate anions, Kleinberg and his research team developed a multi-pronged approach to counter plaque acids, promote sustained alkali production and support remineralization and healthy microbial ecosystems on tooth surfaces. By combining these nutrients in a tasty sugar free soft chew, BasicBites mimics how saliva interacts with the oral microbiome to support enamel health.
Ingredients and Evidence of their Efficacy
Arginine
Numerous studies have clearly established that salivary arginine enables various commensal oral bacteria to readily produce acid-reducing alkali. Studies have shown that high levels of free arginine in saliva are associated with caries resistance.(7) These beneficial pH-raising bacteria contain a series of enzymes that constitute the arginine deiminase pathway (ADS), which is of critical importance in oral biofilm pH homeostasis and microbial ecology. Arginine metabolism by the ADS produces alkali in the form of ammonia that counteracts the effects of biofilm acidification. (8)
The many commensal arginolytic species known to express the ADS include Streptococcus sanguinis, Streptococcus gordonii, Streptococcus parasanguis, Streptococcus mitis, Streptococcus oralis, Streptococcus rattus, and certain Lactobacillus species.(9) Arginine metabolism has also been shown to significantly increase plaque pH in vitro and in vivo, even in the presence of carbohydrates.
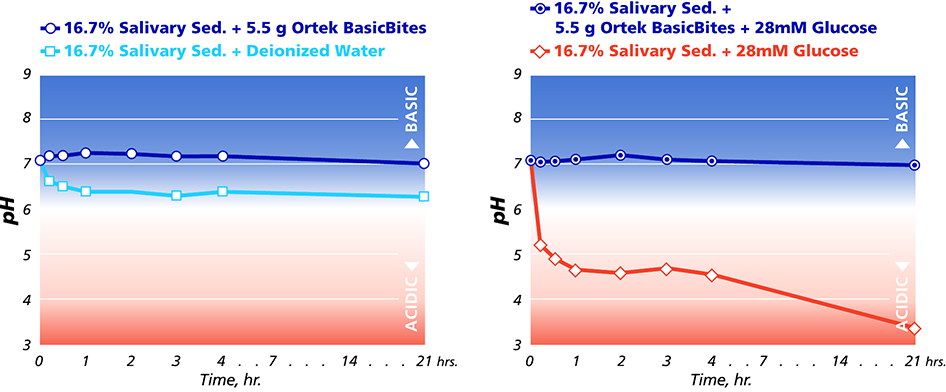
In an in vitro study conducted at the Stony Brook University School of Dental Medicine, BasicBites performed significantly better than the control in sustaining dental plaque pH levels at or near neutrality, even after the introduction of sugar. (10)
It’s been suggested that the anti-caries effect from novel arginine-based oral care technologies is due, in large part, to the ability of arginine to serve as an inducer of the ADS and a substrate for ammonia production by plaque bacteria. (11)
Arginine also has been shown to negatively impact the growth, pathogenic potential, and tolerance of environmental stresses in a way that likely compromises the ability of S. mutans to cause dental caries and infections.(12) Numerous studies indicate that arginine has other beneficial effects on the ecology of the oral microbiome. For example, an oft-cited study showed that treatment with an arginine-containing dentifrice normalized the oral microbiota of caries-active individuals. Its authors noted that, “arginine has a favorable ecological effect on oral microbiome, and thus represents a promising approach to caries control.” (13)
Calcium
This essential component in BasicBites is well-established as an effective way to inhibit enamel demineralization and support remineralization. It suppresses the solubilization of tooth enamel by mass action and reduces the release of calcium from tooth mineral when attacked by plaque acids.(14) Since calcium precedes the release of phosphate from enamel, dentine and cementum during demineralization,(15) it can be more effective than phosphate in inhibiting this process. Also, when pH levels in the oral microbiome are optimized by BasicBites, more calcium is available to support the remineralization process.
Bicarbonate, carbonate anions:
Arginine and calcium at the pH of the mouth have a positive charge. They are then able to form salts and salt complexes when combined preferably with negatively charged anions, such as bicarbonate and carbonate, that are also cariostatic. These two anions in BasicBites enhance or supplement the beneficial activities of calcium and arginine. For example, such anions act as tooth-protecting buffers while also providing a more favorable alkaline pH for base formation from arginine.(14)
Summary
Just two BasicBites a day deliver next-level oral care by:
• Replenishing and coating enamel with a blend of vital nutrients
• Nourishing beneficial pH-raising bacteria discovered in dental plaque
• Activating the production of tooth protecting buffers
• Providing immediate and sustained neutralization of harmful plaque acids
• Enriching enamel and supporting its remineralization
• Helping maintain a pH-balanced microbial ecosystem that favors oral homeostasis
1. Acevedo AM, et al. A One Year Double Blind Placebo Controlled Clinical Study to Assess Progression of Non-cavitated Occlusal Caries Lesions in First Permanent Molars of Children using a Sugarless BasicBites® soft chew confection. Study sponsored by Ortek Therapeutics. https://www.basicbites.com/wp-content/uploads/2018/09/ClinicalAbstract.pdf
2. Acevedo AM, Machado C, Rivera LE, Wolff M, Kleinberg I. The inhibitory effect of an arginine bicarbonate/calcium carbonate CaviStat-containing dentifrice on the development of dental caries in Venezuelan school children. The Journal of Clinical Dentistry. 2005 ;16(3):63-70. PMID: 16305004. https://europepmc.org/article/med/16305004
3. Petrou I, Heu R, Stranick M, Lavender S, Zaidel L, Cummins D, Sullivan RJ, Hsueh C, Gimzewski JK. A breakthrough therapy for dentin hypersensitivity: how dental products containing 8% arginine and calcium carbonate work to deliver effective relief of sensitive teeth. J Clin Dent. 2009;20(1):23-31. PMID: 19489189. https://pubmed.ncbi.nlm.nih.gov/19489189/
4. Acevedo AM, Montero M, Rojas-Sanchez F, Machado C, Rivera LE, Wolff M, and Kleinberg I. Clinical evaluation of the ability of arginine bicarbonate/calcium carbonate complex in a mint confection to inhibit the development of dental caries in children. J Clin Dent. 2008;19(1):1-8.
5. Kleinberg I, Kanapka JA, Chatterjee R, Craw D, D’Angelo NK, Sandham HG (1979). Metabolism of nitrogen by the oral mixed bacteria. In: Saliva and dental caries. Kleinberg I, Ellison SA, Mandel ID, editors. Washington, DC, and London: Information Retrieval, pp. 357-377.
6. Kleinberg I, Jenkins GN, Chatterjee R, Wijeyeweera L (1982). The antimony pH electrode and its role in the assessment and interpretation of dental plaque pH. J Dent Res 61:1139–1147.
7. Van Wuyckhuyse BC, Perinpanayagam HE, Bevacqua D, Raubertas RF, Billings RJ, Bowen WH, Tabak LA. Association of free arginine and lysine concentrations in human parotid saliva with caries experience. J Dent Res. 1995 Feb;74(2):686-90. doi: 10.1177/00220345950740021001. Erratum in: J Dent Res 1995 Jul;74(7):following 1432. PMID: 7722066. https://pubmed.ncbi.nlm.nih.gov/7722066/
8. Nascimento MM. Potential Uses of Arginine in Dentistry. Adv Dent Res. 2018 Feb;29(1):98-103. doi: 10.1177/0022034517735294. PMID: 29355411; PMCID: PMC5784480. https://pubmed.ncbi.nlm.nih.gov/29355411/
9. Burne RA, Marquis RE. Alkali production by oral bacteria and protection against dental caries. FEMS Microbiol Lett. 2000 Dec 1;193(1):1-6. doi: 10.1111/j.1574-6968.2000.tb09393.x. PMID: 11094270. https://pubmed.ncbi.nlm.nih.gov/11094270/
10. The pH response of the mixed bacteria in salivary sediment with BasicBites™ in the presence and absence of glucose generating acid. Stony Brook University School of Dental Medicine. https://www.basicbites.com/wp-content/uploads/2018/09/BasicBitespHResponseStudy.pdf
11. Nascimento, M.M., Burne, R.A. Caries Prevention by Arginine Metabolism in Oral Biofilms: Translating Science into Clinical Success. Curr Oral Health Rep 1, 79–85 (2014). https://doi.org/10.1007/s40496-013-0007-2 https://link.springer.com/article/10.1007/s40496-013-0007-2#citeas
12. Chakraborty B, Burne RA. Effects of Arginine on Streptococcus mutans Growth, Virulence Gene Expression, and Stress Tolerance. Appl Environ Microbiol. 2017 Jul 17;83(15):e00496-17. doi: 10.1128/AEM.00496-17. PMID:
28526785; PMCID: PMC5514675. https://pubmed.ncbi.nlm.nih.gov/28526785/
13. Zheng, X., He, J., Wang, L. et al. Ecological Effect of Arginine on Oral Microbiota. Sci Rep 7, 7206 (2017). https://doi.org/10.1038/s41598-017-07042-w https://www.nature.com/articles/s41598-017-07042-w#citeas
14. I Kleinberg, A New Saliva-Based Anticaries Composition. Dentistry Today. 1999 Feb;18(2):98-103.
15. Abou Neel EA, Aljabo A, Strange A, Ibrahim S, Coathup M, Young AM, Bozec L, Mudera V. Demineralization-remineralization dynamics in teeth and bone. Int J Nanomedicine. 2016 Sep 19;11:4743-4763. doi: 10.2147/IJN.S107624. PMID: 27695330; PMCID: PMC5034904. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5034904/
Related Clinical Studies
Schiff T, Delgado E, Zhang YP, Cummins D, DeVizio W, Mateo LR. Clinical evaluation of the efficacy of an in-office desensitizing paste containing 8% arginine and calcium carbonate in providing instant and lasting relief of dentin hypersensitivity. Am J Dent. 2009 Mar;22 Spec No A:8A-15A.
Ayad F1, Ayad N, Zhang YP, DeVizio W, Cummins D, Mateo LR. Comparing the efficacy in reducing dentin hypersensitivity of a new toothpaste containing 8.0% arginine, calcium carbonate, and 1450 ppm fluoride to a commercial sensitive toothpaste containing 2% potassium ion: an eight-week clinical study on Canadian adults. J Clin Dent. 2009;20(1):10-6.
Schiff T, Mateo LR, Delgado E, Cummins D, Zhang YP, DeVizio W. Clinical efficacy in reducing dentin hypersensitivity of a dentifrice containing 8.0% arginine, calcium carbonate, and 1450 ppm fluoride compared to a dentifrice containing 8% strontium acetate and 1040 ppm fluoride under consumer usage conditions before and after switch-over. J Clin Dent. 2011;22(4):128-38.
Kraivaphan P, Amornchat C, Triratana T, Mateo LR, Ellwood R, Cummins D, DeVizio W, Zhang YP. Two-year caries clinical study of the efficacy of novel dentifrices containing 1.5% arginine, an insoluble calcium compound, and 1450ppm fluoride. Caries Res. 2013;47(6):582-90. doi: 10.1159/000353183. Epub 2013 Aug 28.
Yin W1, Hu DY, Fan X, Feng Y, Zhang YP, Cummins D, Mateo LR, Pretty IA, Ellwood RP. A clinical investigation using quantitative light-induced fluorescence (QLF) of the anticaries efficacy of a dentifrice containing 1.5% arginine and 1450 ppm fluoride as sodium monofluorophosphate. J Clin Dent. 2013;24 Spec no A:A15-22.
Yin W1, Hu DY, Li X, Fan X, Zhang YP, Pretty IA, Mateo LR, Cummins D, Ellwood RP. The anti-caries efficacy of a dentifrice containing 1.5% arginine and 1450 ppm fluoride as sodium monofluorophosphate assessed using Quantitative Light-induced Fluorescence (QLF). J Dent. 2013 Aug;41 Suppl 2:S22-8. doi: 10.1016/j.jdent.2010.04.004.
Srisilapanan P1, Korwanich N, Yin W, Chuensuwonkul C, Mateo LR, Zhang YP, Cummins D, Ellwood RP. Comparison of the efficacy of a dentifrice containing 1.5% arginine and 1450 ppm fluoride to a dentifrice containing 1450 ppm fluoride alone in the management of early coronal caries as assessed using Quantitative Light-induced Fluorescence. J Dent. 2013 Aug;41 Suppl 2:S29-34. doi: 10.1016/j. jdent.2010.04.005.
Hu DY1, Yin W, Li X, Feng Y, Zhang YP, Cummins D, Mateo LR, Ellwood RP. A clinical investigation of the efficacy of a dentifrice containing 1.5% arginine and 1450 ppm fluoride, as sodium monofluorophosphate in a calcium base, on primary root caries. J Clin Dent. 2013;24 Spec no A:A23-31.
Souza MLR, Cury JA, Tenuta LMA, Zhang YP, Mateo LR, Cummins D, et al. Comparing the efficacy of a dentifrice containing 1.5% arginine and 1450 ppm fluoride to a dentifrice containing fluoride alone in the management of primary root caries. Journal of Dentistry. 2013;41S:35-41
Cantore R, Petrou I, Lavender S, Santarpia P, Liu Z, Gittins E, Vandeven M, Cummins D, Sullivan R, Utgikar N. In situ clinical effects of new dentifrices containing 1.5% arginine and fluoride on enamel de- and remineralization and plaque metabolism. J Clin Dent. 2013; 24 Spec no A:A32-44.
Wolff M, Corby P, Klaczany G, Santarpia P, Lavender S, Gittins E, Vandeven M, Cummins D, Sullivan RJ. In vivo effects of a new dentifrice containing 1.5% arginine and 1450 ppm fluoride on plaque metabolism. Clin Dent. 2013;24 Spec no A:A45-54.
Santarpia RP III, PHD, Lavender S, PHD, Gittens E, BS, Vandeven M, PHD, Cummins D, PHD, Sullivan R, PHD. A 12-week clinical study assessing the clinical effects on plaque metabolism of a dentifrice containing 1.5% arginine, an insoluble calcium compound and 1,450 ppm fluoride. American Journal of Dentistry, Vol. 27, No, 2, Apr 2014
Li X, Zhong Y, Jiang X, Hu Deyu, Mateo LR, Morrison BM Jr, Zhang YP. Randomized clinical trial of the efficacy of dentifrices containing 1.5% arginine, an insoluble calcium compound and 1450 ppm fluoride over two years. J Clin Dent. 2015;26(1):7-12.
Additional Supporting Scientific Data:
Kleinberg I, Jenkins G.
The pH of plaques in the different areas of the mouth before and after meals and their relationship to the pH and flow of resting saliva.
Archs Oral Biol. 1964;9:493-516.
Kleinberg I, Kanapka J, Chatterjee R, et al. Metabolism of nitrogen by the oral mixed bacteria. In: Kleinberg I, Ellison S, Mandel I, eds. Saliva and Dental Caries. New York: Information Retrieval; 1979:357-377.
Wijeyeweera R, Kleinberg I. Acid-base pH curves in vitro with mixtures of pure cultures of human oral microorganisms. Archs Oral Biol. 1989;34:55-64.
Kleinberg I. A mixed-bacteria ecological approach to understanding the role of oral bacteria in dental caries causation: an alternative to streptococcus mutans and the specific plaque hypothesis. Crit. Rev. Oral Biol. Med. 2002;13(2):108-25.
Chatterjee R, Kleinberg I. Bacterial acidification and Cavistat alkalinization of occlusal fissure pH. CD-ROM of Abstracts. J. Dent. Res., Special Issue A., 84: 3264, 2005.
Petrou I, Heu R, Stranick M, Lavender S, Zaidel L, Cummins D, Sullivan RJ, Hsueh C, Gimzewski JK. A breakthrough therapy for dentin hypersensitivity: how dental products containing 8% arginine and calcium carbonate work to deliver effective relief of sensitive teeth. J ClinDent. 2009;20(1):23-31.
M.M. Nascimento,1,* Y. Liu,2 R. Kalra,2 S. Perry,3 A. Adewumi,4 X. Xu,5 R.E. Primosch,4 and R.A. Burne2. Oral Arginine Metabolism May Decrease the Risk for Dental Caries in Children. J Dent Res. 2013 Jul; 92(7): 604–608.
Nascimento MM, Browngardt C, Xiaohui X, Klepac-Ceraj V, Paster BJ, Burne RA. The effect of arginine on oral biofilm communities. Mol Oral Microbiol. 2014 Feb;29(1):45-54. doi: 10.1111/omi.12044. Epub 2013 Dec 2.doi: 10.1371/journal.pone.0121835. eCollection 2015.
Reyes E, Martin J, Moncada G, Neira M, Palma P, Gordan V, Oyarzo JF, Yevenes I. Caries-free subjects have high levels of urease and arginine deiminase activity. J Appl Oral Sci. 2014 Jun;22(3):235-40.
Sharma S, Lavender S, Woo J, Guo L, Shi W, Kilpatrick-Liverman L, Gimzewski JK. Nanoscale characterization of effect of L-arginine on Streptococcus mutans biofilm adhesion by atomic force microscopy. Microbiology. 2014 Jul;160(Pt 7):1466-73. doi: 10.1099/mic.0.075267-0. Epub 2014 Apr 24.
Sullivan R, Rege A, Corby P, Klaczany G, Allen K, Hershkowitz D, Goldder B, Wolff M. Evaluation of a dentifrice containing 8% arginine, calcium carbonate, and sodium monofluorophosphate to repair acid-softened enamel using an intra-oral remineralization model. J Clin Dent. 2014;25(1 Spec No A):A14-9.
Koopman JE, Röling WF, Buijs MJ, Sissons CH, ten Cate JM, Keijser BJ, Crielaard W, Zaura E. Stability and resilience of oral microcosms toward acidification and Candida outgrowth by arginine supplementation. Microb Ecol. 2015 Feb;69(2):422-33. doi: 10.1007/s00248-014-0535-x. Epub 2014 Nov 30.
Huang X, Schulte RM, Burne RA, Nascimento MM. Characterization of the arginolytic microflora provides insights into pH homeostasis in human oral biofilms. Caries Research: 2015;49(2):165-76. doi: 10.1159/000365296. Epub 2015 Jan 28.
Zheng X, Cheng X, Wang L, Qiu W, Wang S, Zhou Y, Li M, Li Y, Cheng L, Li J, Zhou X, Xu X. Combinatorial effects of arginine and fluoride on oral bacteria. J Dent Res. 2015 Feb;94(2):344-53. doi: 10.1177/0022034514561259. Epub 2014 Dec 4.
Kolderman E, Bettampadi D, Samarian D, Dowd SE, Foxman B, Jakubovics NS, Rickard AH. L-arginine destabilizes oral multi-species biofilm communities developed in human saliva. PLoS One. 2015 May 6;10(5):e0121835. doi: 10.1371/journal.pone.0121835. eCollection 2015.
He J1, Hwang G2, Liu Y2, Gao L2, Kilpatrick-Liverman L3, Santarpia P3, Zhou X4, Koo H5. l-Arginine Modifies the Exopolysaccharide Matrix and Thwarts Streptococcus mutans Outgrowth within Mixed-Species Oral Biofilms. J Bacteriol. 2016 Sep 9;198(19):2651-61. doi: 10.1128/JB.00021-16. Print 2016 Oct 1.
Koopman JE1, Hoogenkamp MA1, Buijs MJ1, Brandt BW1, Keijser BJ2, Crielaard W1, Ten Cate JM1, Zaura E3. Changes in the oral ecosystem induced by the use of 8% arginine toothpaste. Arch Oral Biol. 2017 Jan;73:79-87. doi: 10.1016/j.archoralbio.2016.09.008. Epub 2016 Sep 26.
M. Agnello, L. Cen, N.C. Tran, W. Shi, J.S. McLean, and X. He. Arginine Improves pH Homeostasis via Metabolism and Microbiome Modulation. J Dent Res. 2017 Jul; 96(8): 924–930.
Ruth G. Ledder, Hitesh Mistry, Prem K. Sreenivasan, Gavin Humphreys, Andrew J. McBain. Arginine Exposure Decreases Acidogenesis in Long-Term Oral Biofilm Microcosms. 2017 Aug 23;2(4):e00295-17. doi: 10.1128/mSphere.00295-17.eCollection 2017 Jul-Aug.
Dr. AntarikshyaPrabir Das, Dr. Swadheena Patro, Dr. Ankita Mohanty, Dr Sanjay Miglani. A Broad Review On Arginine And Its Application In Dentistry. European Journal of Molecular & Clinical Medicine ISSN 2515-8260 Volume 08, Issue 2, 2021.
Yudong Liu, Shanshan Liu, Qinghui Zhi, Peilin Zhuang, Rongxiu Zhang, Zhenzhen Zhang, Kai Zhang, and Yu Sun. Arginine-induced metabolomic perturbation in Streptococcus mutans. J Oral Microbiol. 2022; 14(1): 2015166.